Introduction to quantum mechanics
Source: WikipediaNature at the subatomic scale abandons "common sense" for a reality that is fundamentally counterintuitive.
Classical physics perfectly explains the world we see—the path of a baseball or the orbit of the Moon. However, as scientists peered into the subatomic world at the end of the 19th century, these rules broke down. Quantum mechanics emerged to describe this "absurd" reality, where particles don't behave like tiny billiard balls but follow laws that defy everyday experience.
This shift wasn't just a refinement of old ideas but a total paradigm revolution. In the quantum realm, nature is characterized by "strangeness" like entanglement—where two particles remain connected across any distance—and superfluidity, where liquid helium can spontaneously flow up and over the walls of its container. As physicist Richard Feynman famously noted, quantum mechanics describes "nature as she is—absurd."
The "quanta" revolution proved that energy is delivered in discrete packets rather than continuous waves.
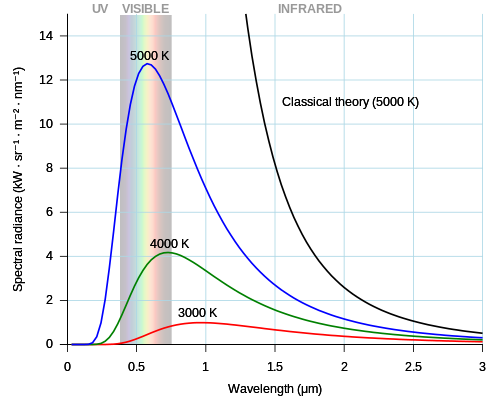
For centuries, light was understood as a continuous wave, similar to ripples in a pond. This model failed to explain "black-body radiation" (the way hot objects glow) until Max Planck suggested that energy is actually divided into individual units called "quanta." Albert Einstein took this further, proposing that light itself consists of "energy quanta" (now called photons), which explained why the speed of ejected electrons in the photoelectric effect depends on the color of light, not its brightness.
This discovery transformed our understanding of matter. By realizing that energy isn't a smooth flow but a series of "chunks," scientists were able to solve inconsistencies that had baffled classical physicists. It proved that at the most fundamental level, the universe is "digital" rather than "analog."
Atoms remain stable because electrons occupy "quantized" orbits rather than spiraling into the nucleus.
Early models of the atom resembled mini-solar systems, with electrons orbiting a nucleus like planets. Classically, these electrons should lose energy and crash into the center, making matter impossible. Niels Bohr solved this by "quantizing" the orbits: electrons can only exist in specific, fixed energy levels. They don't "travel" between these levels; they "jump" instantaneously, absorbing or emitting light at very specific frequencies.
This quantization explains the unique "fingerprints" of light (spectral lines) emitted by different elements. It turned the mystery of atomic structure into a predictable system, forming the basis for modern chemistry. We now understand that atoms interact and form molecules based entirely on these rigid, quantized rules of electron behavior.
Matter possesses a dual identity, behaving as both a solid particle and a spreading wave.
One of the most startling discoveries of the 20th century was that "corpuscles" like electrons aren't just solid dots; they also move as waves. Louis de Broglie proposed that all matter has a wavelength, a theory later proven when electrons were fired at targets and produced diffraction patterns—a behavior previously thought unique to light and water waves.
This "wave-particle duality" is best seen in the double-slit experiment. A single particle fired at two slits seems to pass through both simultaneously as a wave of probability, only "settling" into a single position once it hits a detector. This suggests that the very act of measurement or observation forces nature to choose a specific state, a concept known as complementarity.
Heisenberg’s Uncertainty Principle reveals a fundamental limit to what can be known about the universe.
In classical physics, if you have a good enough sensor, you can measure a car's position and speed with perfect accuracy. Werner Heisenberg proved this is physically impossible for subatomic particles. The more precisely you know a particle's position, the less precisely you can know its momentum, and vice-versa.
This isn't a failure of our technology; it is a fundamental property of the universe. It implies that particles do not have "defined" traits until they are measured. On a human scale, these uncertainties are too small to notice, but on an atomic scale, they create a "fuzzy" reality where probability replaces the absolute certainty of classical laws.